The ideal bond angles for this geometry are 109.5°.Īs an example of the tetrahedral structure,the figure at left shows the bond geometry of methane If the SN = 4, the observed structures are based on the tetrahedron.įor the case with all four electron domains bonding (then thereĪre four atoms bonded to the central atom), the positions of theīound atoms lie at the corners of a tetrahedron. Groups (as are all the examples mentioned above) this results in an ideal bond angle of 120°. For the case in which all electron groups are bonding He VSEPR model predicts a trigonal planar geometry, with electron groups oriented toward theĬorners of an equilateral triangle. The organic molecule formaldehyde (CH 2O), The VSEPR model predicts a linear geometry and aĮxamples of trigonal planar geometry (SN = 3) are provided by boron trichloride (BCl 3), That is, in each case they count as one electron group, and In the latter both count the same in determining the steric number SN (i.e. Note that the single bonds in the former and the double bonds Note that any atom obeying the octet rule will not have SN > 4.Īnd we'll begin by analyzing the possible molecular shapes for steric number equal to 2, 3 and 4.Įxamples of linear geometry (SN = 2)are provided by beryllium dichloride (BeCl 2) andĬarbon dioxide (CO 2). The value we assign to SN is the same as the number of electron domains (electron groups). (Tro's "electron groups") instead of electron pair in determining a value for SN. Or triple-bonded to the central atom, we sometimes use the term electron domain Since we end up counting a bonded atom only once, even if it is double. Tro's text refers to this count, what we are calling steric number, The number of lone pairs present on the central atom.
#MOLECULAR GEOMETRY PLUS#
This amounts simply to a count of the number of bonded atoms plus Number (abbreviated SN) for the central atom of a molecular structure.
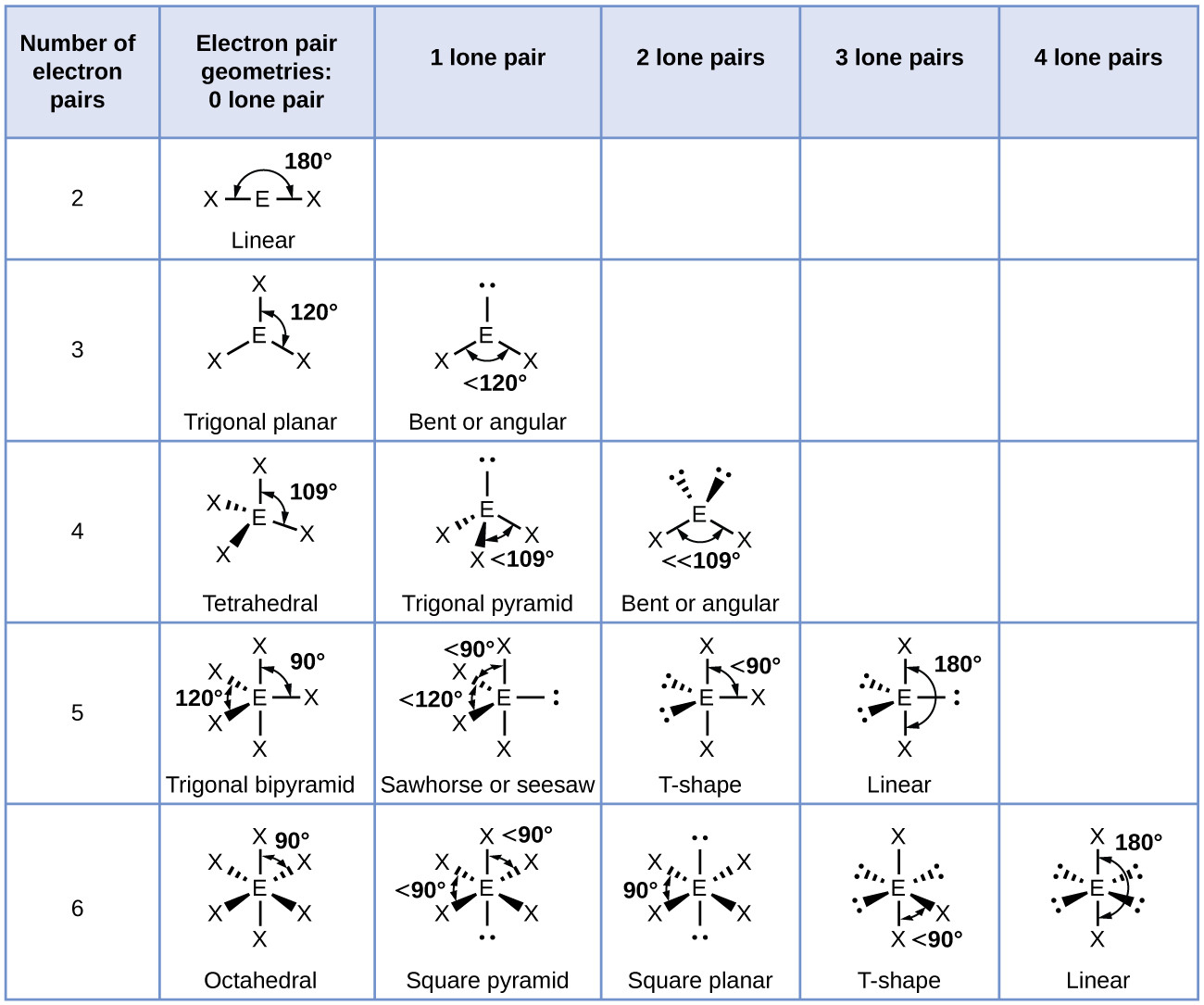
To do this, we apply the valence shell electron pair repulsion or VSEPR model. Shape of the molecule, or its three-dimensional structure. Molecular structure: The five basic shapesįor simple molecules, we can use them to predict the
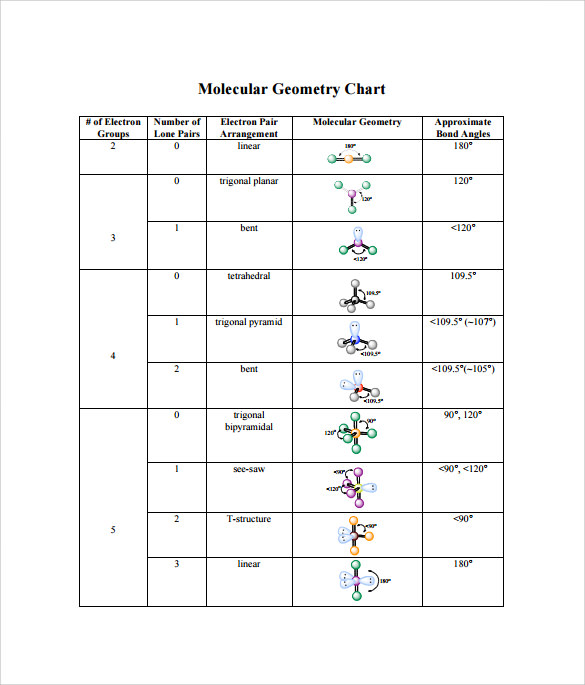
Single-central atom structures: The five basic shapes. Simulated molecules show an excellent agreement with their counterpartsĬomputed using classical quantum chemistry methods.GENERAL CHEMISTRY TOPICS Molecular structure The optimized geometrical parameters for the Simulations are performed using the PennyLane library for quantumĭifferentiable programming. Rotations are selected according to the norm of their gradient. Using an adaptive algorithm where excitation gates in the form of Givens Used to prepare the electronic ground state for each molecule were designed The algorithm is applied to find theĮquilibrium geometries of the $\mathrm$ molecules. Molecule is obtained by minimizing a more general cost function that depends onīoth the quantum circuit and the Hamiltonian parameters, which are Molecule by explicitly considering the parametric dependence of the electronic Variational quantum algorithm for finding the most stable structure of a Impractical already for few-atom systems.
#MOLECULAR GEOMETRY PDF#
Authors: Alain Delgado, Juan Miguel Arrazola, Soran Jahangiri, Zeyue Niu, Josh Izaac, Chase Roberts, Nathan Killoran Download PDF Abstract: Classical algorithms for predicting the equilibrium geometry of stronglyĬorrelated molecules require expensive wave function methods that become
